[最も選択された] pcl5 ba(oh)2 306400-Pcl5+ba(oh)2
Instructions To balance a chemical equation, enter an equation of a chemical reaction and press the Balance button The balanced equation will appear aboveMilk of Magnesia, a suspension of solid Mg(OH)2 in an aqueous medium, is sometimes used to neutralize excess stomach acid Write a complete balanced equation for the neutralization reaction, and identify the conjugate acidbase pairsA) 400 mL b) 333 mL c) 278 mL d) 1 103
Hal Archives Ouvertes Fr
Pcl5+ba(oh)2
Pcl5+ba(oh)2-Balance the following equation H2SO4 Ba(OH)2 → BaSO4 2 H2O If 980 g of H2SO4 reacts, how many grams of BaSO4 should beH 3O OH WORK A NSWER What volume of 00 M H3PO4 is required to neutralize 300 cm3 of 0025 M Ba




Answered A Mixture Of 118 6 G Of Cl2 And 25 9 G Bartleby
Nahso4与ba(oh)2反应求方程式 碱性nahso4 ba(oh)2 = naoh baso4 h2o 中性2nahso4 ba 这些物质在水中都水解sicl4 水解生成硅酸和盐酸、pcl5水解生成磷酸和盐酸 、ca3p2 水解生成氢氧化钙和磷化氢 、mg3c2 水解生成氢氧化镁和甲烷 物质的水解有一定的规律One of relatively few reactions that takes place directly between two solids at room temperature isBa(OH)2 • 8H2O(s) NH4SCN(s) → Ba(SCN)2(s) H2O(l) NH3(g)In this equation, the • 8H2O in Ba(OH) 2 • 8H2O indicates the presence of eight water molecules This compound is called barium hydroxide octahydratebThis program was created with a lot of help from The book "Parsing Techniques A Practical Guide" (IMHO, one of the best computer science books ever written);
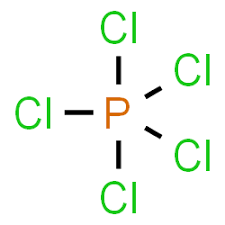
The PCl5 structure has 2 different kinds of PCl bonds All the PhosphorusChlorine equatorial bonds make 90 degrees and 1 degrees bond angles, two each, with the further bonds in the atom The second type of bond is the axial bond Each of these bonds between P and Cl makes 3 90 degrees and 180 degrees bond angles with the supplementary bonds5 Balance the following equation K2CO3 2 HCl → H2O CO2 2 KCl Determine the theoretical yield (grams) of H2O if you start with 345 g of K2CO3 If only 34 g of H2O is produced, what is the percent yield?Standand Enthalpies of Formation & Standard Entropies of Common Compounds Substance State ∆H f S (kJmol) (Jmol·K) Ag s 0 426 Ag aq 727 AgCl s − 962
A litre solution containing NH 4 Cl and NH 4 OH has hydroxide ion concentration of 106 mol L1 Which of the following hydroxide could be precipitated when the solution is added to 1L solution of 01M metal ions?Create Phosphorus oxychloride appears as a colorless fuming liquid with a pungent odor Density 140 lb / gal Very toxic by inhalation and corrosive to metals and tissue Used in gasoline additives and hydraulic fluids CAMEO Chemicals Phosphoryl trichloride is a phosphorus coordination entity(1) The equilibrium constant K c for the reaction PCl 3 (g) Cl 2 (g) ↔ PCl 5 (g) is 49 at 230°C If 0700 mol of PCl 3 is added to 0700 mol of Cl 2 in a 100L reaction vessel at 230°C, what is the concentration of PCl 3 when equilibrium has been established?
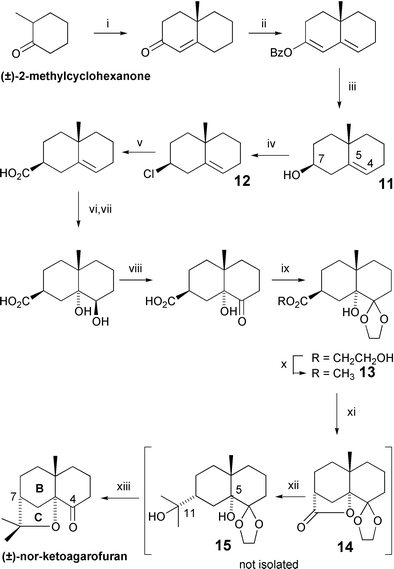



Celastraceae Sesquiterpenoids Biological Activity And Synthesis Chemical Society Reviews Rsc Publishing



Pubs Rsc Org
The Gold Parsing System (Hats off!Na O 2 > Na 2 O Al H 2 SO 4 > Al 2 (SO 4) 3 H 2 O P O 2 > P 2 O 5 Lập phương trình hóa học và cho biết tỉ lệ số nguyên tử, số phân tử của các chất trong phản ứng Bài 2 Đốt cháy bột nhôm trong không khí thu được nhôm axit (Al 2 O 3) a) Viết phương trình hóa họcName Date Balancing Equations About Chemistry http//chemistryaboutcom Balance the following chemical equations 1 Fe 2 3 H 2S0 4
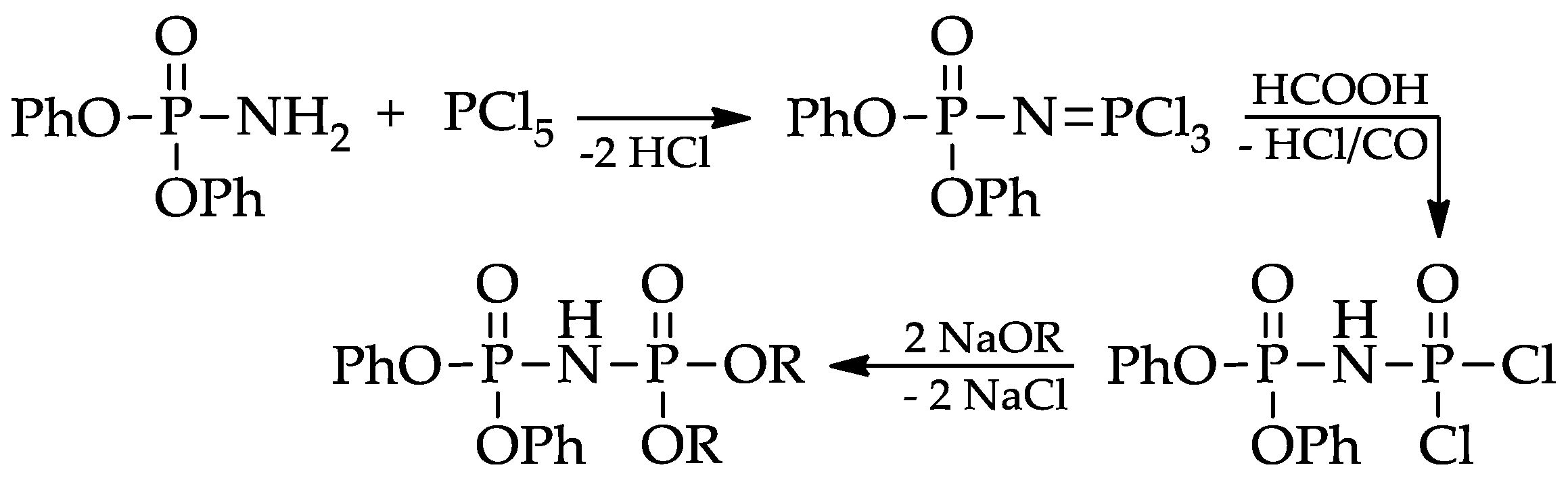



Applied Sciences Free Full Text Linear 2 Ethylhexyl Imidophosphoric Esters As Effective Rare Earth Element Extractants Html




What Is Poh Of 0 02 M Solution Of Ca Oh 2 Youtube
Balancing Equations Practice Problems 1 Balance each of the following equations (a) Fe Cl2 −−→ FeCl3 (b) Fe O2 −−→ Fe2O3 (c) FeBr3 H2SO4 −−→ Fe2(SO4)3 HBr (d) C4H6O3 H2O −−→ C2H4O2 (e) C2H4 O2 −−→ CO2 H2O (f) C4H10O O2 −−→ CO2 H2O (g) C7H16 O2 −−→ CO2 H2O (h) H2SiCl2 H2O −−→ H8Si4O4 HCl (i) HSiCl3 H2O −−SO2 PCl5 —> SOCl2 POCl3 1625g 2378g ?g 1625g SO2 x (1 mol SO2 / 6406g SO2) x (1 mol SOCl2 / 1 mol SO2) x (116g SOCl2 / 1 mol SOCl2) = 30CO2 adalah senyawa kovalen non polar 4 Tata Nama Senyawa Berilah nama senyawasenyawa berikut menurut aturan IUPAC 1MgO = Magnesium Oksida 2SCl6 = Sulfur Heksaklorida 3CuSO4 = Cu2 SO42 ® Tembaga (II) Sulfat 4K2MnO4 = Kalium Permanganat 5N2O4 = Dinitrogen Tetraoksida




13 Pcl5 Acirc Dagger Oelig Pcl3 Cl2 Agrave Reg Agrave Reg Copy Agrave Macr Agrave




Solved Consider The Unbalanced Chemical Equation Below Pc15 Chegg Com
HCOOH PCl5 → HCOCl POCl3 HCl Công dụng của Axit Formic – HCOOH Được sử dụng như một hỗn hợp với axit xitric hoặc HCl vì một mình nó không có khả năng loại bỏ cặn oxit sắt Cân bằng phản ứng Na2SO4 Ba(OH)2 ra gì (và phương trình (NH4)2SO4 Ba(OH)2)Ba(OH) 2 1 H 2 SO 4 1 9808 BaSO 4 1 H 2 O 2 1802 Units molar mass g/mol, weight g Please tell about this free chemistry software to your friends!Aluminum Al(s) 0 2 0 AlCl 3 (s)7042 Al 2 O 3 (s) Aqueous Solutions Ca 2 (aq) CO 3 2(aq)



Pneumatiek Com



1
Chemical bond A chemical bond is a lasting attraction between atoms, ions or molecules that enables the formation of chemical compounds The bond may result from the electrostatic force of attraction between oppositely charged ions as inBa(OH) 2 H 2 SO 4 2 H 2 O BaSO 4 Cl 2 2 NaI 2NaCl I 2 P 4 10 Cl 2 4 PCl 5 H 2 O 2 H 2 O 2 D 2 F 3 A 4 C Select the correct statement below that fits a theoretical yield when a reaction between equal masses of Ca and O occurs to produce CaO A The mass of CaO produced equals the sum of the starting masses of Ca and O BTitle Chapter 04 Chemical Reactions Author Matlock, Stephanie M Last modified by tauraichiku Created Date PM Other titles



1



1
2 SO 4 fi 2 NH 3 SO 2 H 2 decomposition b) Br 2 2 KI fi 2 KBr I 2 single replacement c) 2 Na Cl 2 fi 2 NaCl synthesis d) Al(OH) 3 3 HCl fi AlCl 3 3 H 2 O double replacement 2 Balance each of the equations shown below a) 5 C 2 SO 2 fi 1 CS 2 4 CO2(s) −2766 Ca(OH) 2(s) −9866 CuO(s) −1552 Mg(OH) 2(s) −9247 PCl 3(g) −3064 CaCl 2(s) −7950 CuS(s) −485 MgCl 2(s) −6418 PCl 5(g) −39 CaCO 3(s) −170 CuSO 4(s) −7699 MgCO 3(s) −1113 SiO 2(s) −8594 CaO(s) −6355 Fe 2O 3(s) −22 MgO(s) −6018 SnCl Question # The energy diagram shown represents the chemical reaction between solid ammonium chloride and solid barium hydroxide octahydrate 2NH4Cl (s)Ba (OH)2⋅8H2O (s)→2NH3 (aq)BaCl2 (aq)10H2O (l) The ΔH for this reaction is 548 kJ




Part I Iii Key




Phosphorous Pentachloride Pcl5 Grade Standard Chemical Grade For Industrial Id
Ba(ClO 3) 2 (s) ‐ ‐ Ba(ClO 4) 2•3H 2O (s) ‐ ‐ Ba(IO 3) 2 (s) ‐ ‐864 Ba(IO 3) 2•H 2O (s) ‐ ‐ Ba(N 3) 2•H 2O (s) ‐306 1 ‐ Ba(NO 3) 2 (s) ‐997 ‐ Ba(OH) 2•8H 2O (s) ‐ ‐ Ba(ReO 4) 2•4H9 A 5124g sample of Ba(OH) 2 is dissolved in enough water to make 1 liters of solution How many mL of this solution must be diluted with water in order to make 100 liter of 0100 molar Ba(OH) 2?Ba(OH) 2 (Ksp = 5 x 103) Ni(OH) 2 (K sp = 16 x 1016) Mn(OH) 2 (K sp = 2 x 1013) Fe(OH) 2 (K sp = 8 x 1016) I and IV III and IV



Indole And Indazole Derivatives Having A Cell Tissue And Organ Preserving Effect Patent




Equilibrium Systems Are Chapter Pcl3 Cl2 Pcl5 What Is Kc Acirc Euro 15 The Equilibrium Constant
Cl = 2 mol NH 4 OH x 1605 g NH 4 Cl = 107 g NH 4 Cl 3 mol NH 4 OH PLEASE DO NOT WRITE ON THIS TEST PAPER 21 Examine the following balanced reaction 2AlCl 3 3MgSO 4 o Al 2 (SO 4) 3 3MgCl 2 2 mol 3 mol 1 mol If you have 8 moles of AlCl 3 and 9 moles of MgSO 4, which is your limiting reactant?A) H 2 O B) CH 3 OH C) CH 3 CH 2 OH D) HF E) NaF Ans E 22 PCl5 (g) ⇄ PCl3 (g) Cl2 (g) Find the molarity of the Ca(OH)2 solution if mL of acid are required to reach the equivalence point H3O OH WORK ANSWER What volume of 075 M H2SO4 is required to neutralize 250 mL of 0427 M KOH?



Neyco Fr




Polymerization Mechanism Of 1 3 Benzoxazine Catalyzed By Pcl5 And Rearrangement Of Chemical Structures Sciencedirect
Times New Roman Comic Sans MS Diseño predeterminado Formuleu Àc sulfúric Òxid de mercuri(II) Hidròxid de cobalt ( III ) Heptaòxid de diiode Òxid de plom (II) Hidròxid de berilli SO3 PCl5 Ba(OH)2 NaH Co2O3 Triclorur de fòsfor silà Sulfur d'amoni Diòxid de sofre Iodur d'hidrogen PbO2 I2O5 Be(OH)2 HCl CaS Ió alumini Ió hidròxidWhen these compounds dissolve in water, hydroxide ions are released directly into the solution For example, KOH and Ba(OH) 2 dissolve in water and dissociate completely to produce cations (K and Ba 2, respectively) and hydroxide ions, OH – These bases, along with other hydroxides that completely dissociate in water, are considered strongDirect link to this balanced equation Instructions on balancing chemical equations Enter an equation of a chemical reaction and click 'Balance' The answer will




How To Balance Ba Oh 2 H3po4 Ba3 Po4 2 H2o Barium Hydroxide Phosphoric Acid Youtube



Deltaplus Eu
Science Chemistry Chemistry questions and answers Classify each of these reactions Pb (NO_3)_2 NiCl_2 rightarrow PbCl_2 Ni (NO_3)_2 combination decomposition single replacement double replacement Pb (OH)_2 rightarrow PbO H_2O combination decomposition single replacement double replacement CaO H_2O rightarrow Ca (OH)_2 combinationBa(OH) 2 F 2 PCl 3 The chemical formulas for covalent compounds are referred to as molecular formulas because these compounds exist as separate, discrete molecules Typically, a molecular formula begins with the nonmetal that is closest to the lower left corner of the periodic table,Balance The Equation NaOH H3PO4 = Na3PO4 H2O 1 Label Each Compound With a Variable a NaOH b H 3 PO 4 = c Na 3 PO 4 d H 2 O 2 Create a System of Equations, One Per Element Na 1 a 0b = 3 c 0d O 1 a 4 b = 4 c 1 d H 1 a 3 b = 0c 2 d P 0a 1 b = 1 c 0d 3 Solve For All Variables a = 3 b = 1 c = 1 d = 3 4
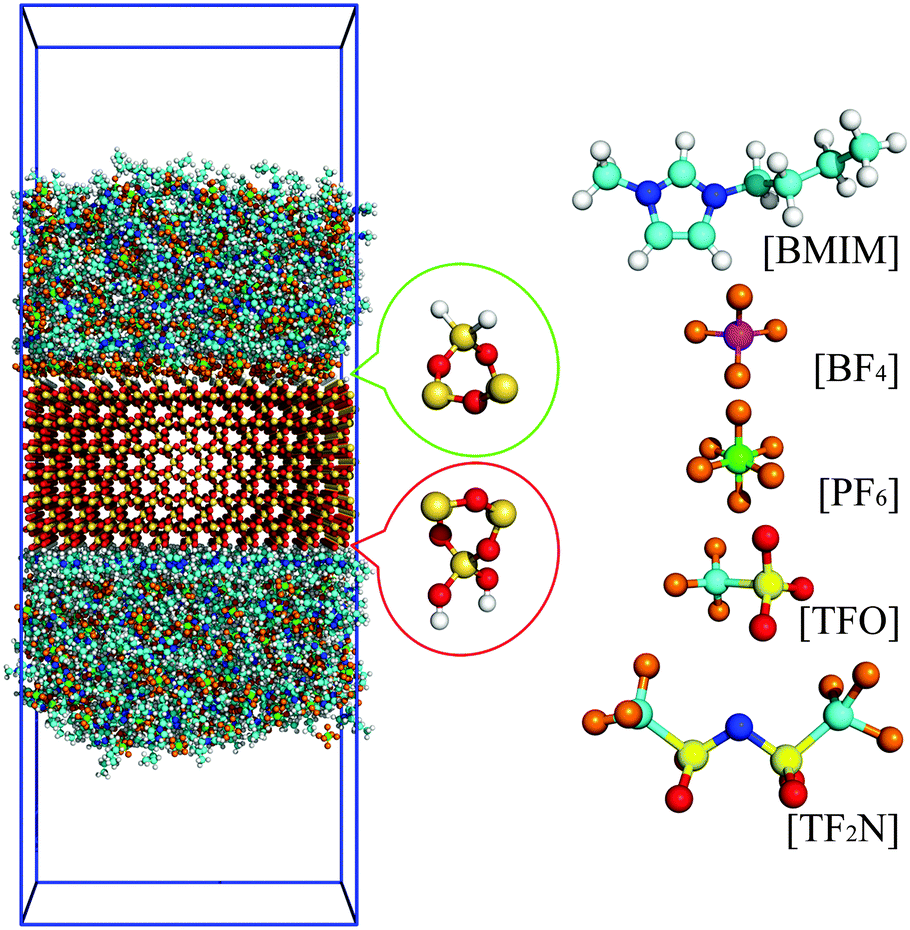



Interfacial Structure And Orientation Of Confined Ionic Liquids On Charged Quartz Surfaces Physical Chemistry Chemical Physics Rsc Publishing Doi 10 1039 C4cpj



Phosphorus Pentachloride Decomposes According To This Equation Pcl5 G Equilibrium Reaction Arrow Pcl3 G Cl2 G An Equilibrium Mixture In A 500 L Flask Chemistry Topperlearning Com 8w5dmudd
2 AgI 1 Na2S 1 Ag2S 2 NaI 2 1 Na3PO4 3 HCl 3 NaCl 1 H3PO4 3 1 Ba3N2 6 H2O 3 Ba(OH)2 2 NH3 4 1 TiCl4 2 H2O 1 TiO2 4 HCl 5 3 CaCl2 2 Na3PO4 1 Ca3(PO4)2 6 NaCl 6 2 NaBr 1 Cl2 2 NaCl 1 Br2 7 1 Mg(OH)2 2 HCl 1 MgCl2 2 H2 O 8 4 FeS 7 O2 2 Fe2O3 4 SO2 9 1 PCl5 4 H2O 1 H3PO4 5 HCl 10 1 C2H6O 3 O2 2 CO2 32HCl(aq) Ba(OH)2(aq) → BaCl2 (aq) 2H2O(l) ∆H = 118 kJCalculate the heat when 1000 mL of 0500 M HCl is mixed with 3000 mL of 0100 M Ba(OH)2 Assuming that the temperature of both solutions was initially 250oC and that the final mixture has a mass of 4000 g and specific heatBa(NO3)2 Barium Nitrate Ba(OH)2 Barium Hydroxide BaCl2 Barium Chloride BaCO3 Barium Carbonate BaI2 Barium Iodide BaO Barium Oxide BaO2 Barium Peroxide BaSO4 Barium Sulfate PCl5 Phosphorus Pentachloride PF3 Phosphorus Trifluoride PH3 Phosphine PI3 Phosphorus Triiodide Pt(NH3)2Cl2 Diamminedichloroplatinum Ra(OH)2 Radium Hydroxide



Solved 1 Convert Each Quantity To An Amount In Moles 4 27 X 10 27 Atoms Of He 2 91 X 10 23 Formula Units Of Ba Oh 2 Ba Oh 2 3 Marks 2 The Calcu Course Hero




Answered A Mixture Of 118 6 G Of Cl2 And 25 9 G Bartleby
Ba(OH)2(s) 2 HNO3(aq) → Ba(NO3)2(aq) 2 H2O(Ú) barium nitric barium water hydroxide acid nitrate 31 Two strong Brönsted acids and one strong Brönsted base Many examples exist Strong acids HCl, HBr, HI, HNO 3 Strong bases LiOH, NaOH, KOH Chapter 3 Chemical Reactions 43 Writing Net Ionic EquationsA stepbystep explanation of how to draw the Ba(OH)2 Lewis Dot StructureFor Ba(OH)2 we have an ionic compound and we need to take that into account when weAnswer Ba (OH)2 ( Barium hydroxide ) is an ionic bond What is chemical bond, ionic bond, Molecular bond?



Indole And Indazole Derivatives Having A Cell Tissue And Organ Preserving Effect Patent



At Technic Com
A 0108 M b 0296 M c 0592 M d 08 M please tell how to identify this things, if its memorization can give you all listBa(OH)2 Sodium Chloride mg cah2 Cu(NO3)2 Barium Chloride ammonium Ca(NO3)2 CaI2 Iron III Oxide no3 Magnesium Phosphate salt Calcium Phosphate Fe(NO3)3 Ca3(PO4)2 Ba(ClO4)2 Barium Fluoride NH4NO2 NH4ClO3 (NH4)2CO3 NaOCH3 CoCl2 MgF2 Rb2O BaCl2 LiF CaF2 Mg3N2 CaBr2 BaO KF CaO PCl5 dinitrogen pentoxide N2O5 CCL4 P4 phosphorus trichlorideE Ba(OH)2 dan CO2 Ba(OH)2 adalah senyawa ion ;
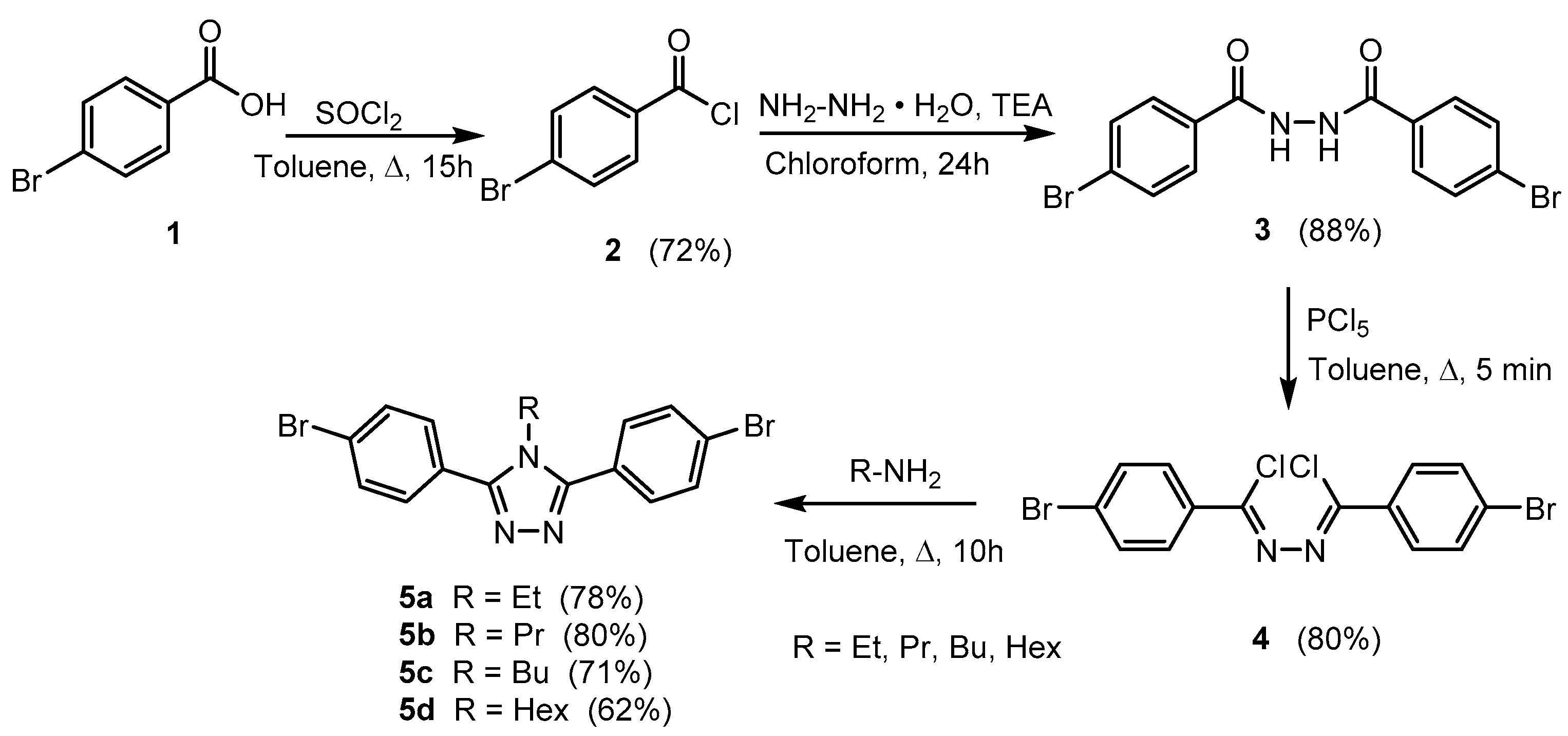



Materials Free Full Text Highly Luminescent 4h 1 2 4 Triazole Derivatives Synthesis Molecular Structure And Photophysical Properties Html




Chemistry An Atoms First Approach 2nd Edition By Steven S Zumdahl Test Bank By Melvinjone Issuu
Phosphorus pentachloride is manufactured by either batch or continuous processing In the former, the phosphorus trichloride usually dissolves in carbon tetrachloride before being treated with chlorineA mixture of about one part of phosphorus trichloride to one part of carbon tetrachloride is introduced to a waterjacketed vessel that contains an efficient stirrer and a tight cover with a3 Ca(OH)2 2 H3PO4 ( Ca3(PO4)2 3 H2O 2 Al(C2H3O2)3 3 Mg(OH)2 ( 2 Al(OH)3 3 Mg(C2H3O2)2 4 FeS 7 O2 ( 2 Fe2O3 4 SO2 Ca3P2 6 H2O ( 3 Ca(OH)2 2 PH3 Sb2S3 6 HCl ( 2 SbCl3 3 H2S 4 Li O2 ( 2 Li2O 92 Practice Problems Already Balanced (synthesis) NH3 HCl ( NH4Cl (decomposition) CaCO3 ( CaO CO2 (synthesis) BaO H2O ( BaCHEM 1411 – STUDYGUIDEforTEST2 (CHAPTERS 4, 5 and 6) 1 Which of the following compounds is a strong electrolyte?
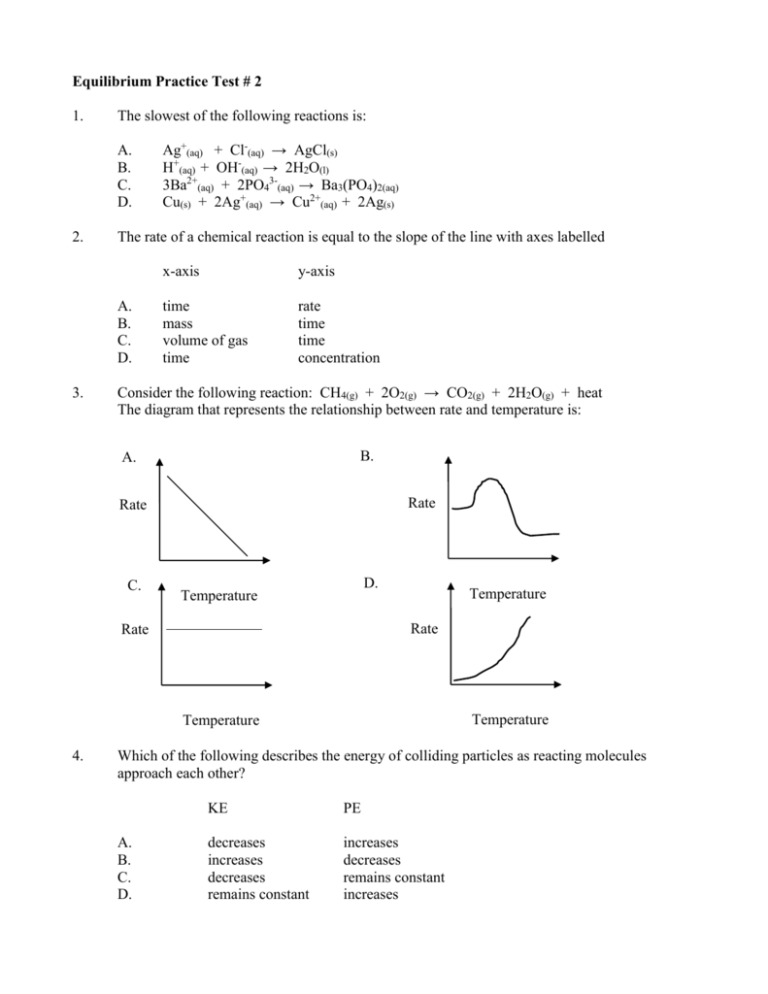



Equilibrium Practice Test 2 Answer Key
2.gif)



Chem 112 Lecture Spring 01 Overheads
What a great software product!) The Calitha GOLD engine (c#) (Made it possible for me to do this program in C#) In a constantpressure calorimeter, 500 mL of 0300 M Ba(OH)2 was added to 500 mL of 0600 M HCl The reaction caused the temperature of the solution to rise from 2173 °C to 25 °C If the solution has the same density and ChemistryBa(OH) 2·8H 2O (s) 8104 BaCl 2 (s) 321 1226 BaSO 4 (s) 1322 Ba(NO 3) 2 (s) 997 2138 Beryllium Be (s) 0 0 950 Be2 (aq) 38 1297 BeO (s) 6096 50 1297 BeCl 2 (s) 4904 4456 68 Bismuth Bi (s) 0




16 Chemistry Morotuwa
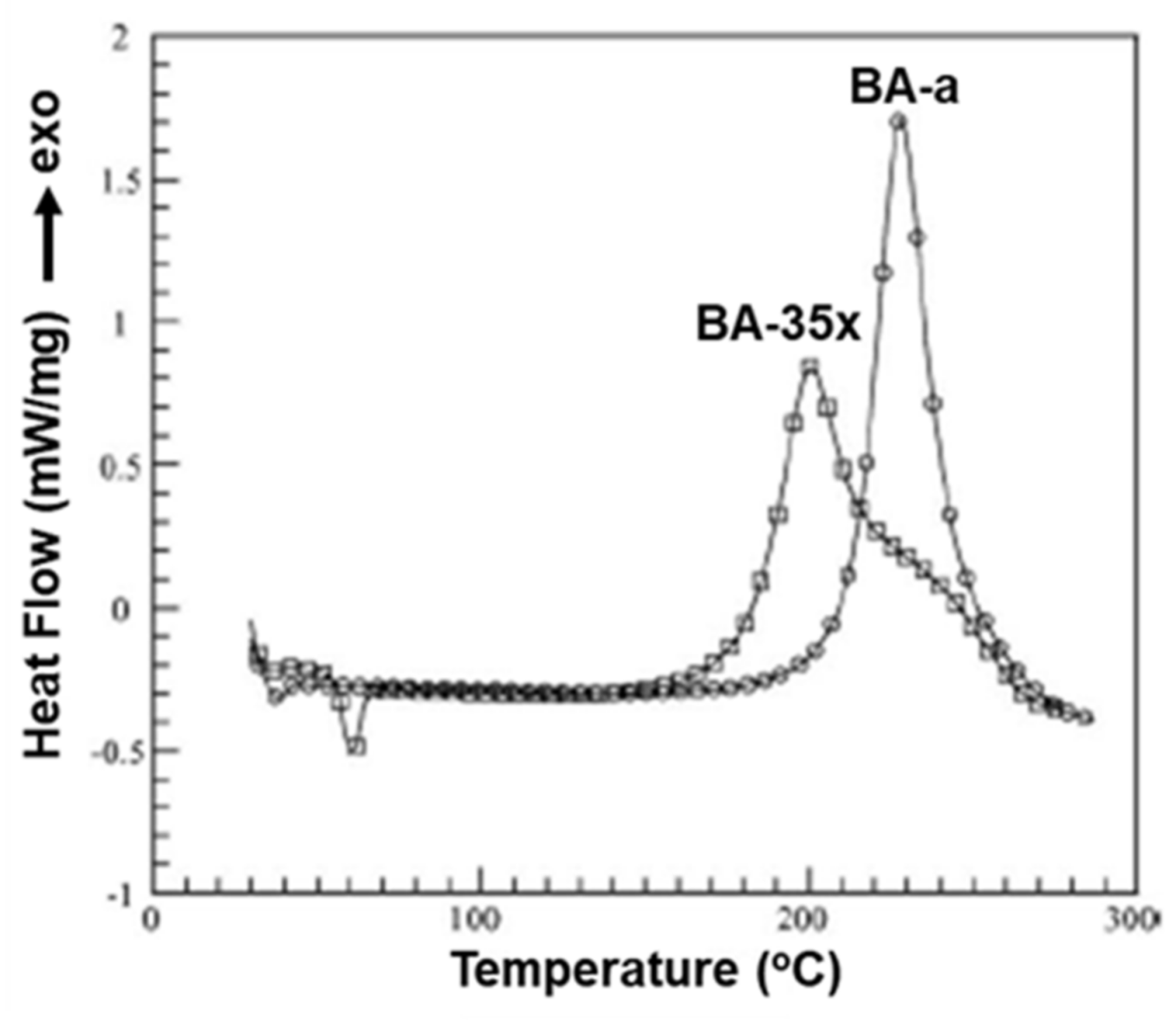



Polymers Free Full Text Review On The Accelerated And Low Temperature Polymerization Of Benzoxazine Resins Addition Polymerizable Sustainable Polymers Html




Structure And Properties Of Slow Resorbing Nanofibers Obtained By Co Axial Electrospinning As Tissue Scaffolds In Regenerative Medicine Peerj




Bhartiyambhartiya Bhartiya With Your Class Section Rollno And Name Pcl5 H2o Acirc Dagger Rsquo H3po4 Hcl V Ba3n2 H2o Acirc Dagger Rsquo Ba Oh 2 Nh3 2 Pdf Document



Formationeda Com



Hal Archives Ouvertes Fr




Substrate Specificity Of Cytolase Pcl5 For Platycosides Download Scientific Diagram




Answered How Many Chlorine Atoms Are There In Bartleby



Theses Fr




Chemistry Solutions Chemistry Solutions 9701 11 M J 14




New Chalcones And Thiopyrimidine Analogues Derived From Mefenamic Acid Microwave Assisted Synthesis Anti Hiv Activity And Cytotoxicity As Antileukemic Agents
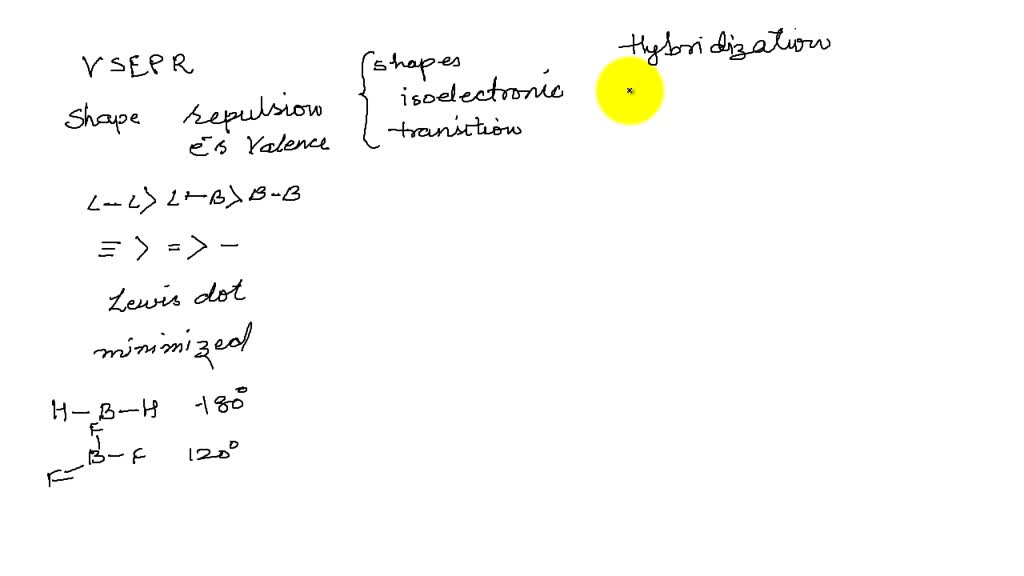



Solved Compare And Contrast The Hybridization And Geometry Of Pcl5 With The Hybridization And Geometry Of Ch4




Pdf Optical Electrical And Electrochemical Properties Of Pcl5 Ito Transparent Conductive Films Deposited By Spin Coating Materials For Single Layer Devices



Solved 1 Convert Each Quantity To An Amount In Moles 4 27 X 10 27 Atoms Of He 2 91 X 10 23 Formula Units Of Ba Oh 2 Ba Oh 2 3 Marks 2 The Calcu Course Hero




Solved Please Help I Just Woke Up And The Deadline Is Chegg Com



Pcl3 Cl2 Pcl5 Balance




13 Pcl5 Acirc Dagger Oelig Pcl3 Cl2 Agrave Reg Agrave Reg Copy Agrave Macr Agrave




Nomenclature And Formula Writing Write The Formula For The



Indole And Indazole Derivatives Having A Cell Tissue And Organ Preserving Effect Patent



Solved The Equilibrium Constant K For The Following Reaction Is 4 68a 10 2 At 536 K Pcl5 G Pcl3 G Cl2 G An Equilibrium Mixture Of The Three Gases In A 17 5 L Container At 536 K




Poly ϵ Caprolactone Functionalized Carbon Nanofibers By Surface Initiated Ring Opening Polymerization Wang 07 Journal Of Applied Polymer Science Wiley Online Library




Polymerization Mechanism Of 1 3 Benzoxazine Catalyzed By Pcl5 And Rearrangement Of Chemical Structures Sciencedirect



Profweb Ca




Pdf Optical Electrical And Electrochemical Properties Of Pcl5 Ito Transparent Conductive Films Deposited By Spin Coating Materials For Single Layer Devices
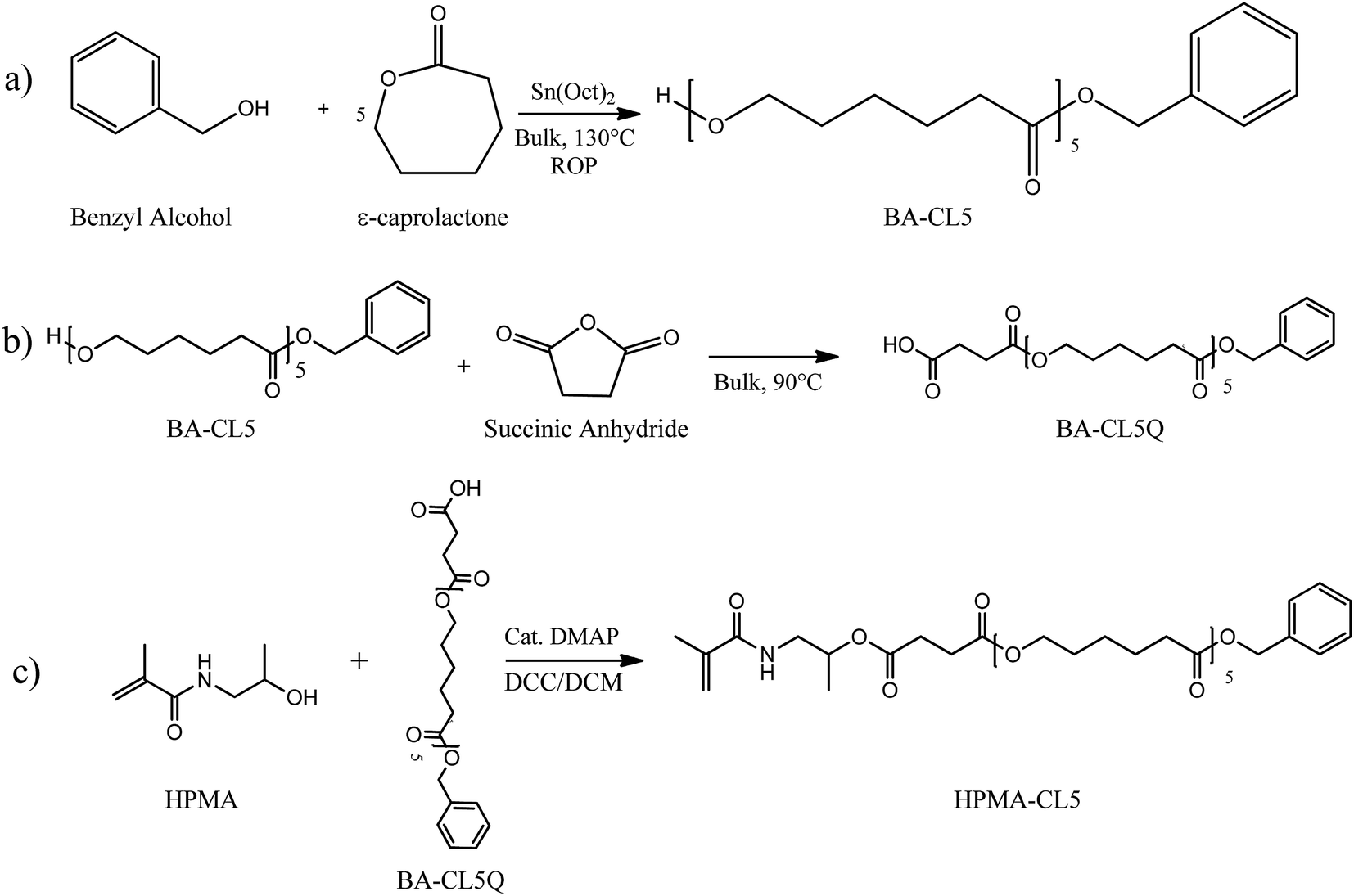



Poly Hpma Based Copolymers With Biodegradable Side Chains Able To Self Assemble Into Nanoparticles Rsc Advances Rsc Publishing




Chemical Equilibrium For Iit Jee




Naming Compounds There Are Three Steps Involved In Naming Ionic Compounds Naming The Cation Naming The Anion And Naming The Entire Compound Pdf Free Download



Hal Archives Ouvertes Fr




Sequential Nucleophilic Arylation Ring Contractive Rearrangement Of N Alkoxylactams Organic Letters




25 Solved Questions On Freshman Chemistry I Test 2 Chem 1211 Docsity
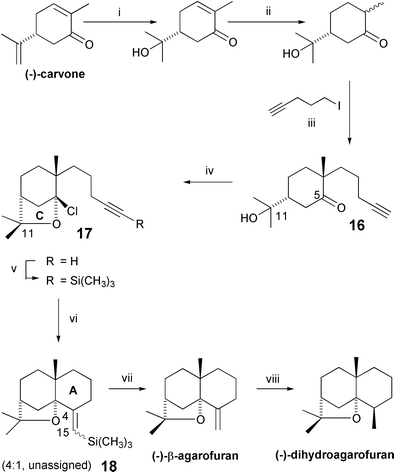



Celastraceae Sesquiterpenoids Biological Activity And Synthesis Chemical Society Reviews Rsc Publishing
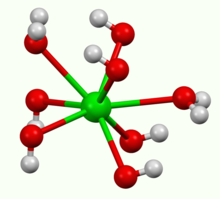



Barium Hydroxide Wikipedia




Magnetsandmotors Litetube




Vdocuments Site Chemistry a633f0



11c Write The Net Ionic Equation Caco3 S H2so4 Aq Caso4 S Co2 G H2o L Youtube




Revision Ecris Le Bon Nom Pour Chaque Formule Chimique Ppt Video Online Telecharger




Bhartiyambhartiya Bhartiya With Your Class Section Rollno And Name Pcl5 H2o Acirc Dagger Rsquo H3po4



Tel Archives Ouvertes Fr



Hal Archives Ouvertes Fr




Optical Electrical And Electrochemical Properties Of Pcl5 Ito Transparent Conductive Films Deposited By Spin Coating Materials For Single Layer Devices Sciencedirect




Sequential Nucleophilic Arylation Ring Contractive Rearrangement Of N Alkoxylactams Organic Letters




Answered 3 Consider The Titration Ba Oh 2 Aq Bartleby




Revision Ecris Le Bon Nom Pour Chaque Formule Chimique Ppt Video Online Telecharger
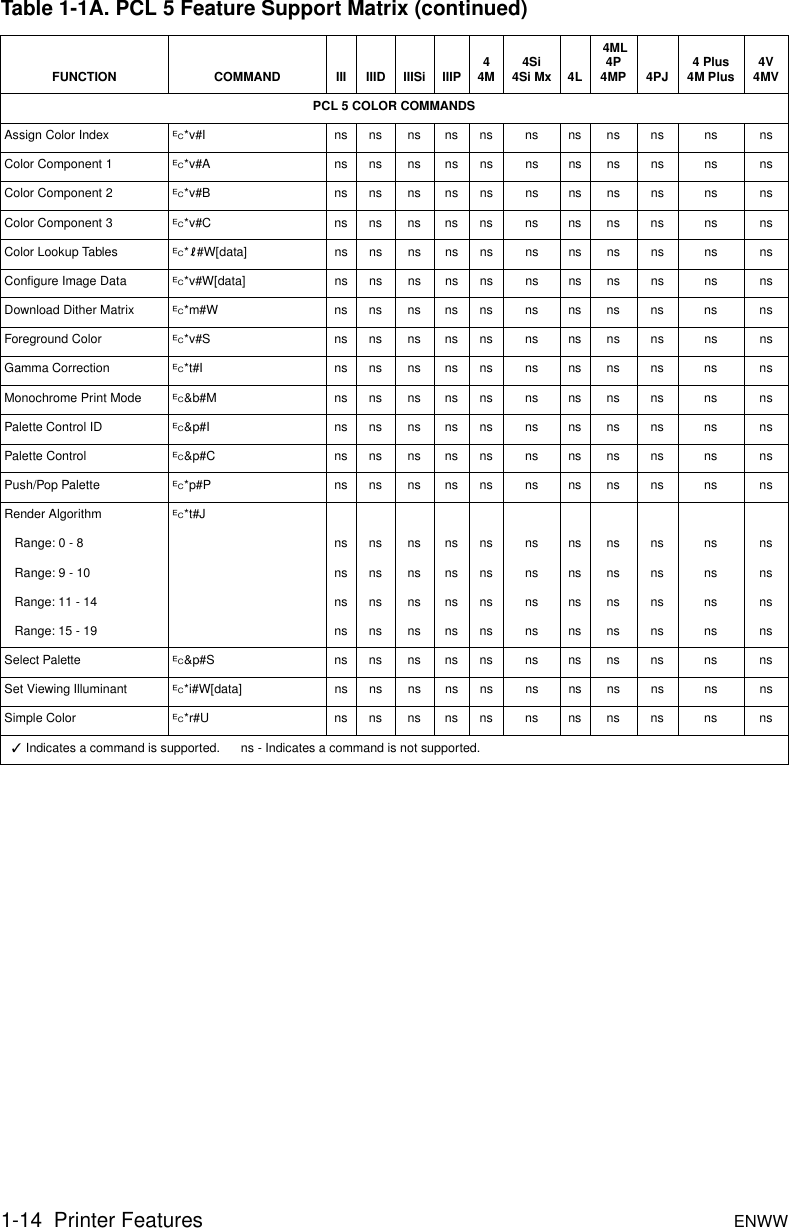



Hp Pcl 5 Comparison Guide Enww Pcl Pjl Reference Bpl136




A Sample Of Pure Pcl5 Was Introduced Into Organic Chemistry



So2 Pcl5 Socl2 Pcl3o Balanced Chemical Equation




Pcl5 Lewis Structure How To Draw The Lewis Structure For Pcl5 Youtube




Epb1 Procede Pour La Preparation De Tamsulosine Et Ses Derives Google Patents




Polymerization Mechanism Of 1 3 Benzoxazine Catalyzed By Pcl5 And Rearrangement Of Chemical Structures Sciencedirect




Pdf Conversion Of Glycosylated Platycoside E To Deapiose Xylosylated Platycodin D By Cytolase Pcl5




Polymerization Mechanism Of 1 3 Benzoxazine Catalyzed By Pcl5 And Rearrangement Of Chemical Structures Sciencedirect




Phosphorus Pentachloride Decomposes According To This Equation Pcl5 G Equilibrium Reaction Arrow Pcl3 G Cl2 G An Equilibrium Mixture In A 500 L Flask Chemistry Topperlearning Com 8w5dmudd




Which Compound Contains Both Covalent And Ionic Bonds 2 Pts A Pcl5 B Cscl C Mgcl2 Homeworklib




The Cylinder Is 15 0 Cm Tall Calculate The Radius 2 0cm Ppt Download



Chemistry Problems Equilibrium Docsity




Decomposition Du Pentachlorure De Phosphore Superprof



Which Chemical Equation Is Balanced A 2pcl5 2h2o 2hcl H3




Solved Which Of The Following Is A Strong Electrolyte Ch3 Cl Nacl Ch3 Ch2 Oh Pcl5




Answered A Mixture Of 118 6 G Of Cl2 And 25 9 G Bartleby



Rasstavte Koefficienty V Uravneniyah Ukazhite K Kakomu Tipu Oni Otnosyatsya 1 Ba No3 2 Al2



What Is The Reaction Product Of Acetamide With P2o5 Quora




Pdf Conversion Of Glycosylated Platycoside E To Deapiose Xylosylated Platycodin D By Cytolase Pcl5




Phosphorus Pentachloride Wikipedia
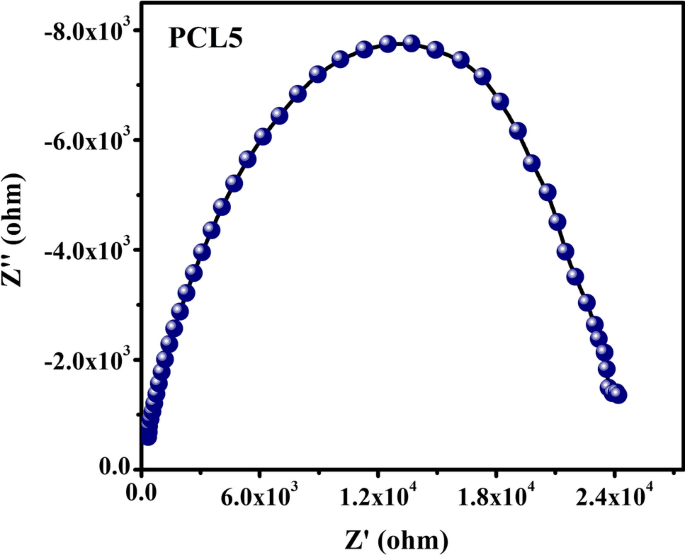



Influence Of Co 2 On The Structure Conductivity And Electrochemical Stability Of Poly Ethylene Oxide Based Solid Polymer Electrolytes Energy Storage Devices Springerlink




Advanced Organic Synthesis Novasep




Answered A Mixture Of 118 6 G Of Cl2 And 25 9 G Bartleby




Epa1 Chiral Phosphoramidimidates And Derivatives Thereof Google Patents
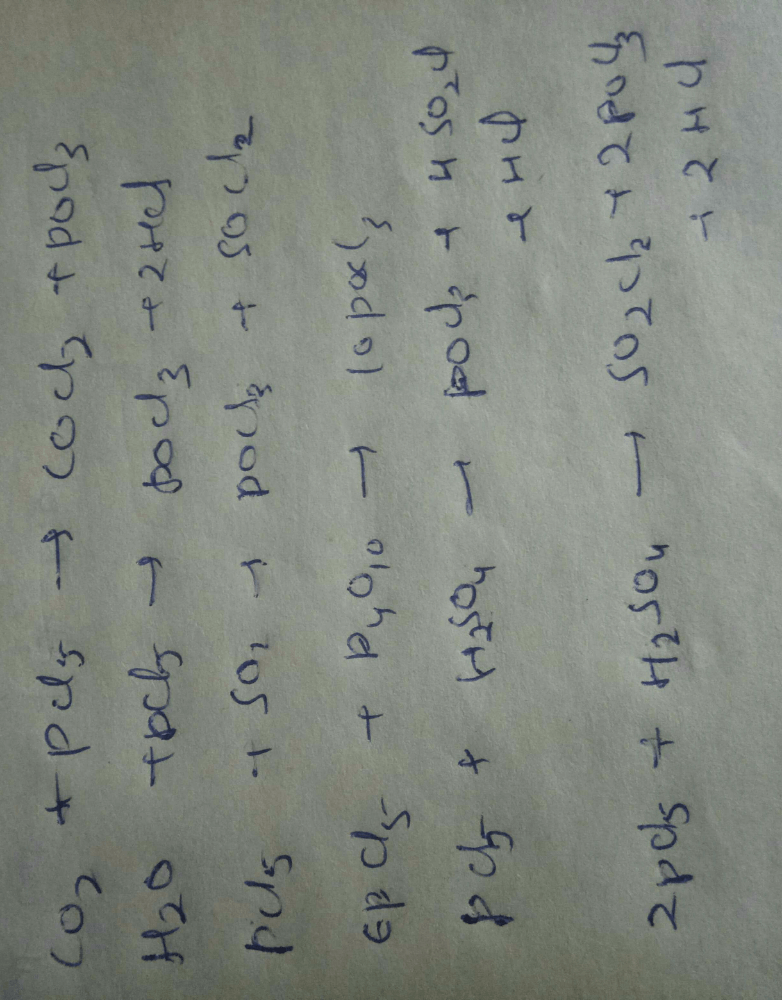



Among The Following The Number Of Compounds Than Can React With Pcl5 To Give Pocl3 Iso2 Co2 So2 H2o H2so4 P4o10correct Answer Is 5 Can You Explain This Answer Edurev Jee




Optical Electrical And Electrochemical Properties Of Pcl5 Ito Transparent Conductive Films Deposited By Spin Coating Materials For Single Layer Devices Sciencedirect
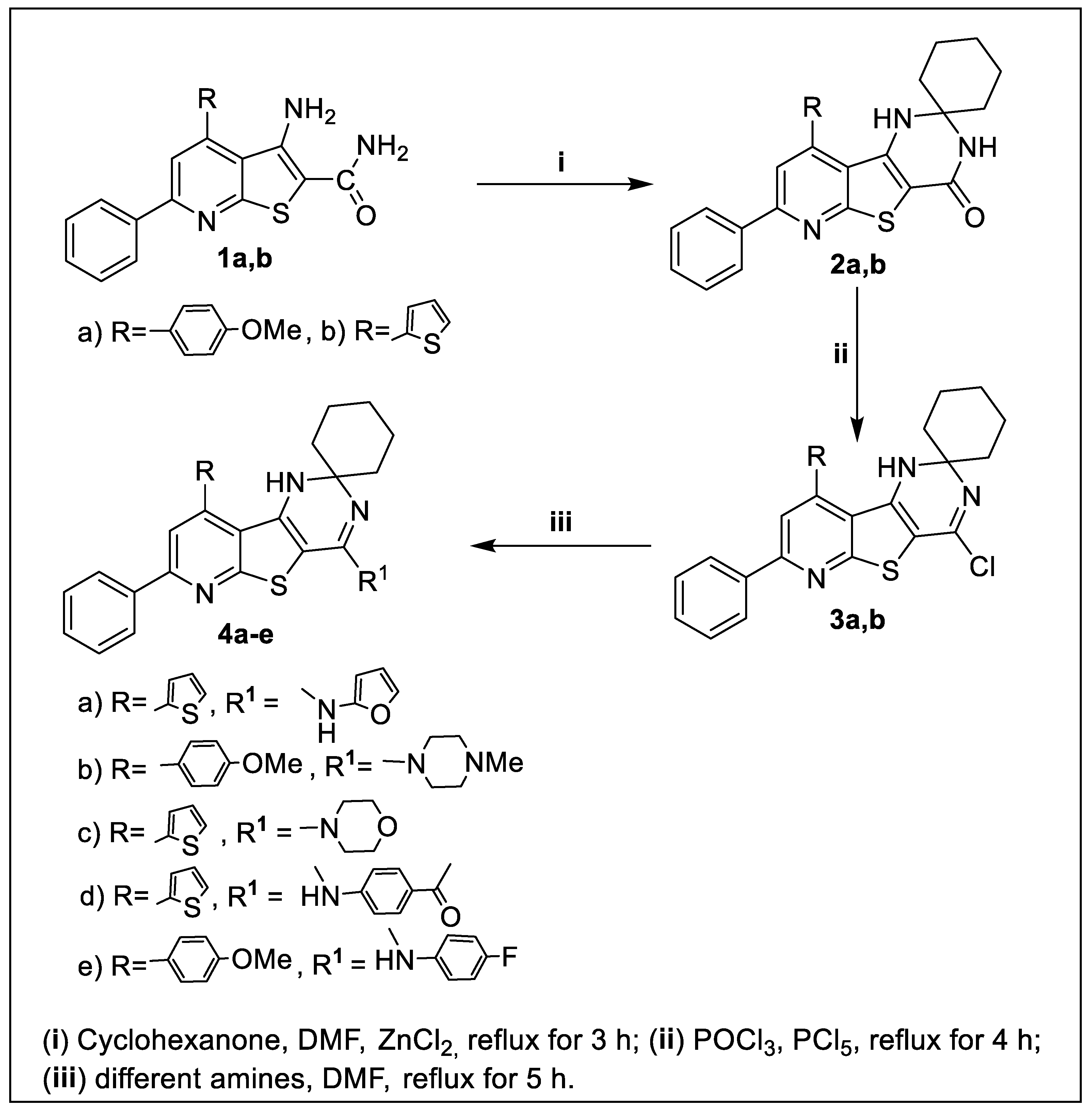



Antibiotics Free Full Text Synthesis And Biological Evaluation Of New Pyridothienopyrimidine Derivatives As Antibacterial Agents And Escherichia Coli Topoisomerase Ii Inhibitors Html




What Is Simplified In This Picture Ppt Download




Equilibrium Systems Are Chapter Pcl3 Cl2 Pcl5 What Is Kc Acirc Euro 15 The Equilibrium Constant




Revision Ecris Le Bon Nom Pour Chaque Formule Chimique Ppt Video Online Telecharger




Basic Chemistry Muhammad Rahim Id 5d1527a74b237




Solved 53 Agi Na2s Ag2s Nal 54 Na Po Hcl Nacl Chegg Com
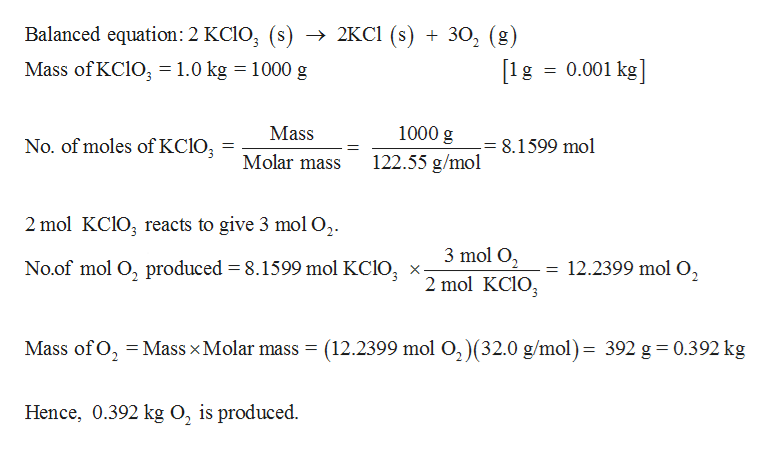



Answered Be Sure That All Of The Chemical Bartleby
Comments
Post a Comment